This is Issue #82 of our In-Depth Science Series on the Folate Industry, dedicated to unpacking the technology, compliance, and risk-avoidance logic behind folate ingredients. Our goal: help businesses choose the right raw materials and avoid costly mistakes; help consumers select truly effective folate without overspending.
After 19 years in folate ingredient development, I’ve seen too many people get it wrong from the start: consumers complain, “I took folate for over six months, but my levels didn’t budge,” while manufacturers worry, “We added active folate, yet batch-to-batch potency fluctuates and products become commoditized, forcing us into a price war.”
The root cause isn’t user error or poor execution — it’s a failure to grasp what each technological leap in folate’s century-long evolution actually solved, and a lack of clarity about the fundamental differences among folate types on the market.
This article lays out the foundational logic of folate in one go. Whether you’re a consumer choosing a product or a business selecting raw materials, you can apply this immediately. Recommended reading and saving.

1-Minute Core Concept Primer (No More Confusion, No More Pitfalls)
Let’s clarify four essential concepts so everything that follows clicks into place and you won’t be misled by marketing jargon:
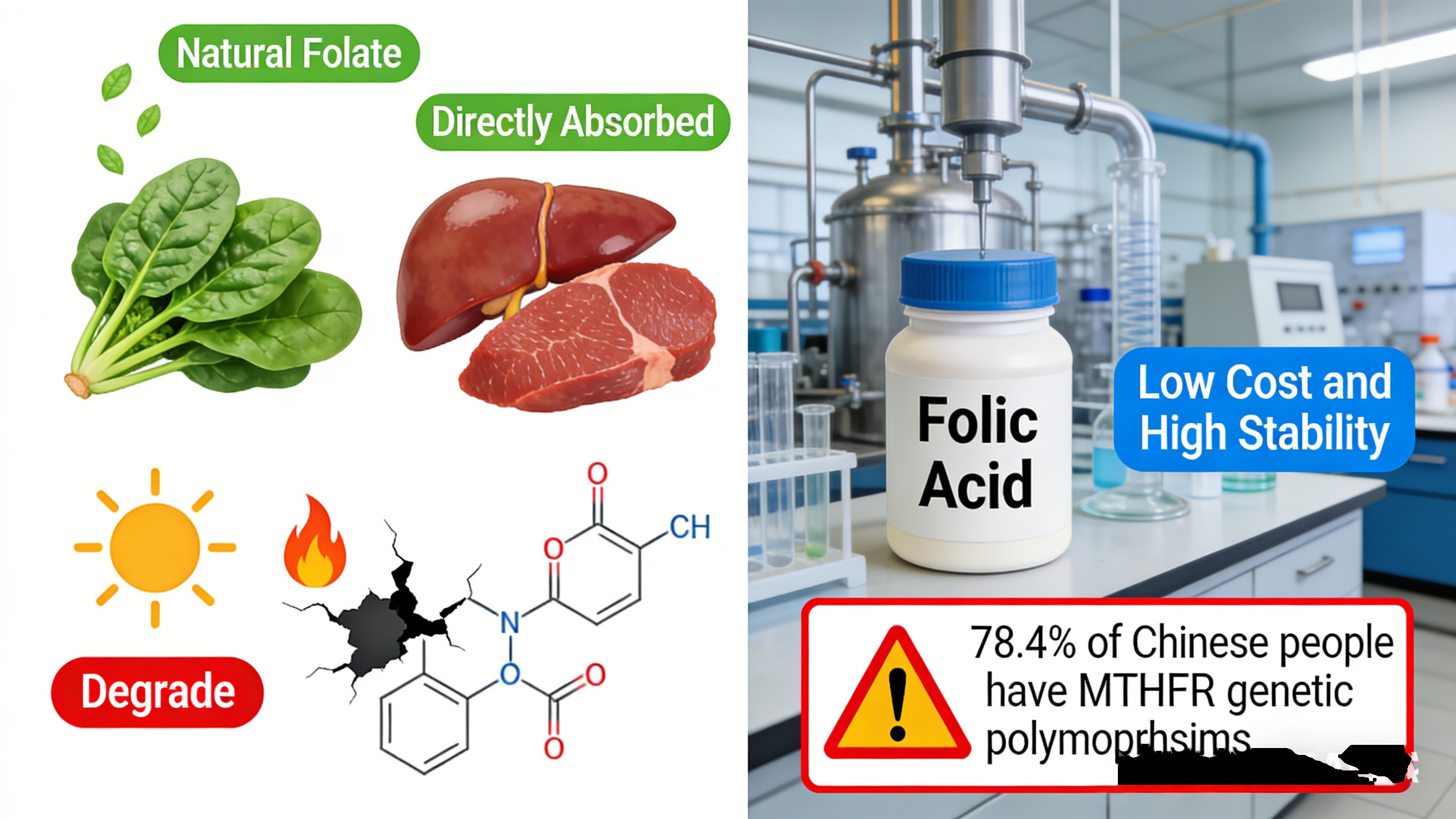
Folate: The naturally occurring active form found in spinach, liver, and other foods. It’s directly absorbable, but degrades rapidly when exposed to light or heat, making standardized mass production impossible.
Folic Acid: A synthetic oxidized folate. Low cost and highly stable, but biologically inactive until converted by human enzymes. 78.4% of Chinese people exhibit polymorphisms in genes related to this metabolic pathway.
Proprietary C-Crystal Naturalization Folate: Our globally patented crystal form of active folate and the world’s first Naturalization folate category. Safety approaches that of food-derived folate. Remains stable at room temperature and meets pregnancy–infant grade quality controls far exceeding domestic and international pharmacopeias. Represents the latest upgrade in folate technology.

Three Industrial Revolutions in Folate: Each Leap Redefined Its Underlying Logic
Folate’s history spans three disruptive technological shifts, each addressing the core limitation of the previous generation to meet stricter safety and application demands.
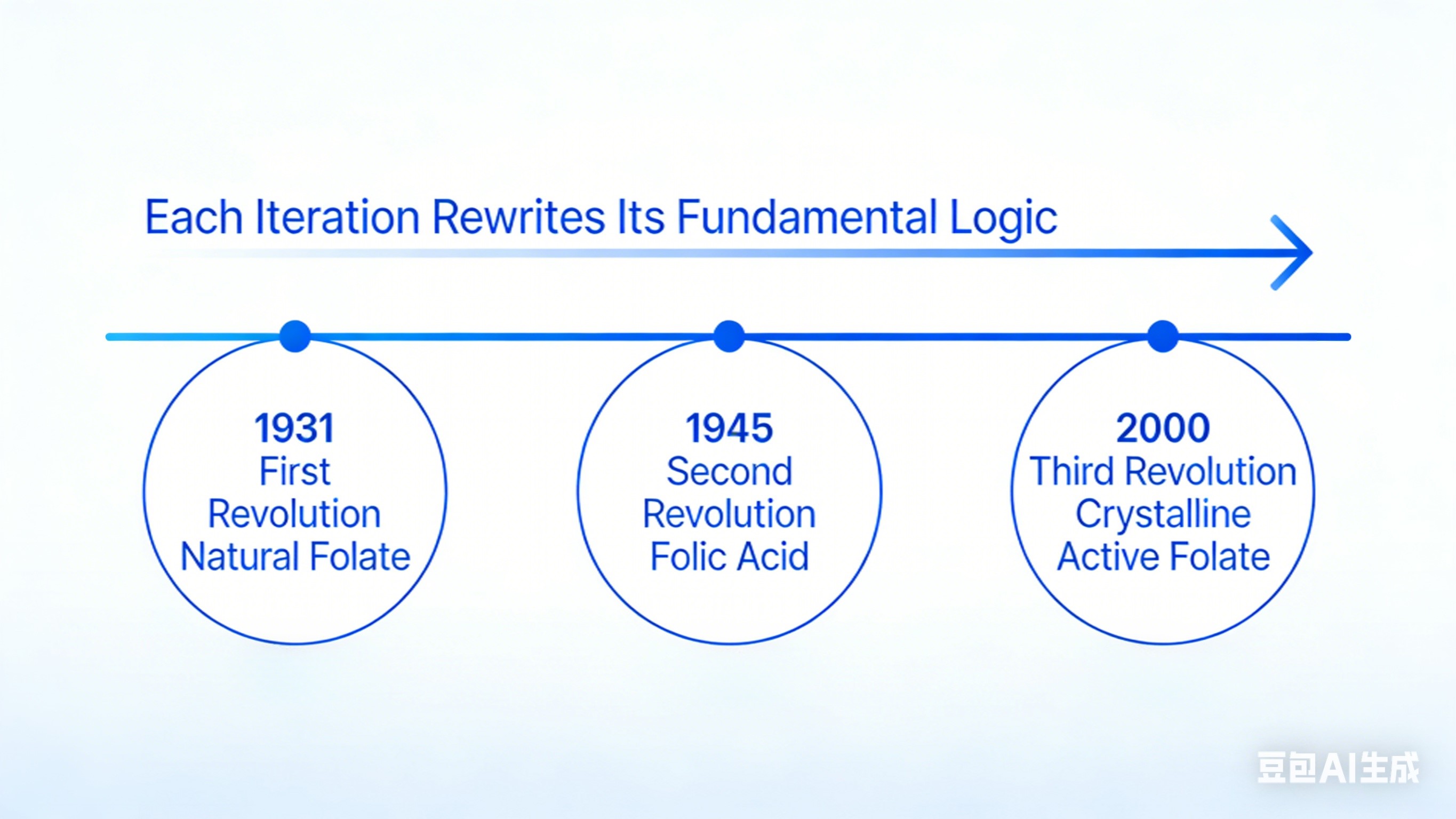
First Revolution: Discovery of Folate and Its Industrial Dead End
The folate story began in 1931, when British physician Lucy Wills discovered that yeast and liver extracts improved anemia in pregnant women — the first identification of this anti-anemia agent. In 1941, American scientists isolated it from spinach leaves and named it “Folate.” By 1943, they extracted milligram quantities of 5-methyltetrahydrofolate — the core active component of natural folate — from tons of spinach.
From birth, however, it was doomed for large-scale civilian use. Chemically unstable, it oxidizes quickly upon exposure to light, heat, or oxygen. Even sealed room-temperature storage can’t guarantee activity retention, ruling out industrial application.
Bottom line: We confirmed natural folate’s nutritional value, but technology couldn’t turn it into a stably produced, standardized product.
Second Revolution: Mass Adoption of Folic Acid and Its Genetic Limitation
In 1945, synthetic pteroylmonoglutamic acid — folic acid — was developed, reshaping the industry. Its oxidized structure is extremely stable, cheap to produce, and scalable, solving natural folate’s mass-production barrier. For half a century, folic acid became the global mainstream; 80 countries and regions mandated its fortification in grains. Most ordinary folate supplements and fortified foods contain folic acid.
Yet ongoing clinical and nutrition research revealed its Achilles’ heel: folic acid lacks intrinsic physiological activity and must undergo four enzymatic steps to become absorbable. My 19 years in R&D show the real-world impact in China: 78% of the population shows MTHFR gene polymorphism, including 28% with severe metabolic impairment — enzyme activity as low as 30% of wild-type individuals.
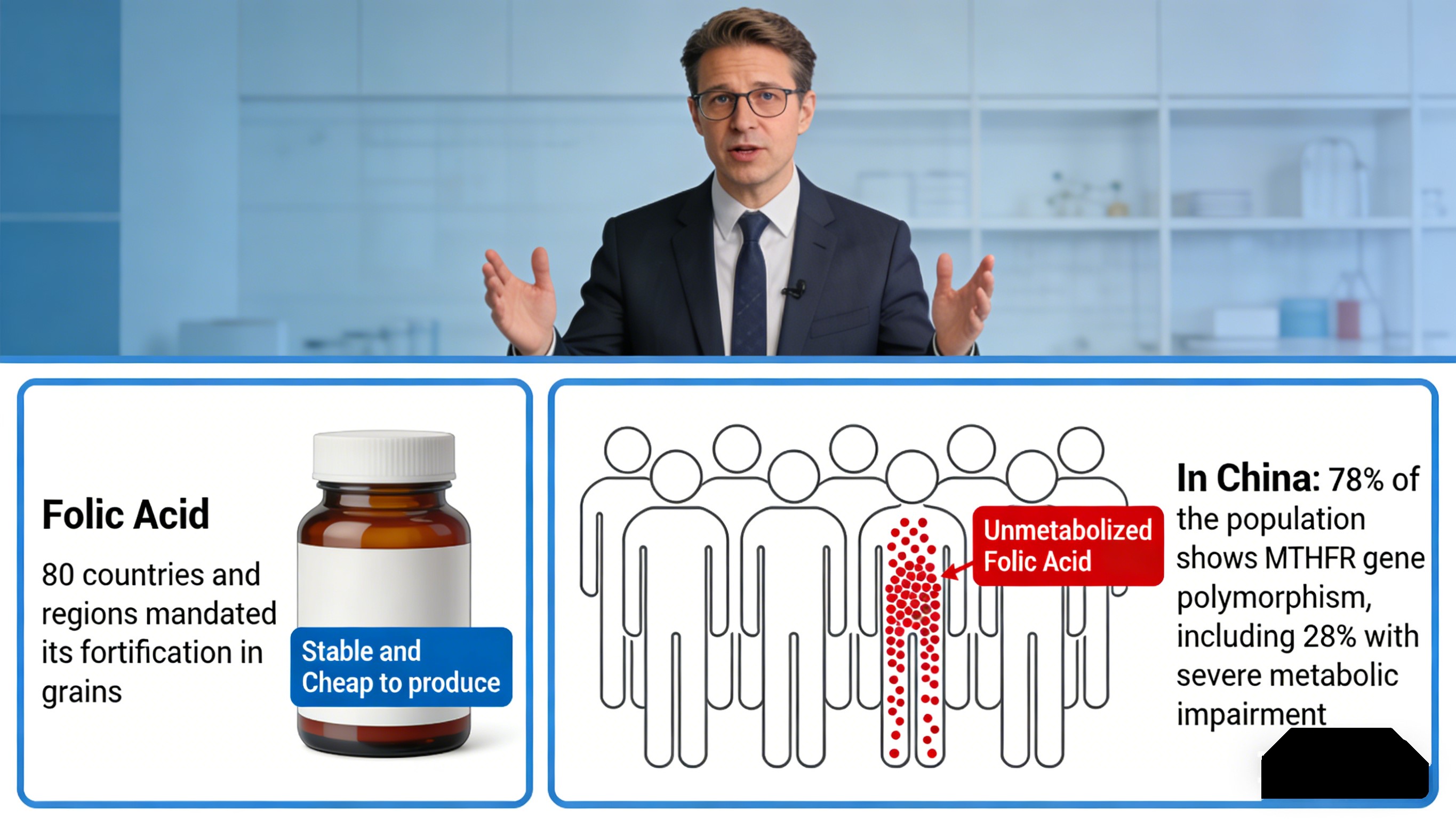
In practical terms, nearly 30% of Chinese people cannot efficiently convert folic acid, leading to ineffective supplementation and potential buildup of Unmetabolized Folic Acid, which may mask vitamin B12 deficiency and interfere with zinc absorption. For companies, this translates into consumer complaints and reputational risk.
Bottom line: Folic acid solved mass production but fails to match the genetic profile of Chinese populations, carrying hidden risks beneath its low cost.
Third Revolution: Breakthrough in Active Folate, Then a New Industry Impasse
In 2000, crystallization technology for active folate achieved a major breakthrough. The first crystalline active folate solved basic stability issues, enabling industrial-scale production of high-purity levorotatory active folate and launching the “active era” of folate.
Active folate’s core advantage: its molecular structure matches that of natural folate’s active component, requiring no metabolic conversion and being directly absorbable — suitable for all populations, including those with folate metabolism disorders.
However, once core patents expired, generic I-crystal active folate flooded the market. Rather than lifting the whole industry, this triggered new problems now faced by 90% of firms:
Homogeneous price wars: With no core technological differentiation, quality varies wildly. Upstream players compete on price, squeezing margins; downstream brands fight over price and traffic, trapped in a vicious cycle.Insufficient quality control for high-safety needs: Some generic producers adhere only to minimum national standards, poorly controlling key impurities. Certain processes still use formaldehyde-based solvents, posing residual toxicity risks, and failing pregnancy–infant grade requirements — creating hidden compliance and reputational traps.
Bottom line: Generic active folate solved metabolic conversion but not industry-wide homogenization or safety ceiling issues.
19 Years of R&D: Our Proprietary C Crystal Creates the Naturalization Folate Era
Our team broke the deadlock through 19 years of focused R&D. When we began in 2007, even unified purity testing standards for active folate were absent domestically; we had to explore every path and regulatory adaptation ourselves. In 2012, we achieved the breakthrough C-crystal form of 6S-5-methyltetrahydrofolate calcium, securing over 40 global patents (e.g., US9150982, CN201410280541.4, EP2805952). We became one of the few companies worldwide with independent IP on active folate crystal forms and the first in China to industrialize it — escaping I-crystal commoditization and opening a new technological track.
Critically, we helped shape China’s active folate regulations, participating in current standard-setting and four rounds of updates. To date, nearly 100 food and maternal–infant nutrition firms in China have launched products using our Naturalization folate, fully compliant and with zero related consumer complaints.
Based on our C-crystal patent, we pioneered the “Naturalization folate” category, earning the first global certification. True Naturalization folate requires meeting three core criteria, clearly distinguishing it from generic I-crystal active folate:

1. Molecular Structure Naturalization: Identical to the folate active component in spinach and other foods. Safety approaches that of dietary folate. Directly absorbed in the gut, covering 100% of users and avoiding ineffective supplementation or Unmetabolized Folic Acid buildup.
2. Crystal Stability Naturalization: Our exclusive C crystal improves thermal and photostability by >30% vs. I crystal, remaining stable at room temperature. No need for cold, dark storage. Near 100% activity retention during processing ensures strong batch-to-batch consistency and minimal potency loss before expiry.
3. Safety Standard Naturalization: Quality controls exceed European and U.S. pharmacopeias: key impurity JK12A ≤0.1%, total impurities ≤0.5%. Synthesis avoids formaldehyde-class solvents, eliminating residue risk. Toxicological studies confirm actual non-toxicity, making it the first active folate in China consistently meeting pregnancy–infant high-safety requirements.
Many peers ask: What can Naturalization folate do for my business? Four tangible values address immediate pain points:
Full Compliance Support: Full dossier, toxicology reports, technical documentation aligned with China’s food additive rules; proven successful launches minimize regulatory risk.Exclusive Differentiation: Global first-mover category certification plus patent licensing create absolute technical distinction, helping capture premium maternal–infant and precision nutrition markets with uncopyable selling points.
End-to-End Cost Control: Domestic production, stable stock, flexible MOQ — 100 g of high-purity Naturalization folate yields ~250,000 tablets (0.4 mg each), costing just a few cents per tablet. Eliminates extra cold storage and material loss expenses.
Zero-Risk Quality Assurance: Ultra-tight impurity control and room-temperature-stable crystal prevent production loss, shelf-life decay, and consumer complaints, slashing compliance and reputational risks.
Three Golden Rules for Folate Selection (Use Immediately, Save for Reference)
Over a century, folate’s three revolutions reflect humanity’s pursuit of safer, more effective, better human-compatible, industrially viable folate forms. Naturalization folate is the upgraded answer.

Whether consumer or business, remember these rules to avoid pitfalls:
1. Priority on Structural Compatibility: Choose 6S-5-methyltetrahydrofolate matching natural folate’s molecular structure — direct absorption, covers all users, prevents inefficacy.2. Crystal Stability as Baseline: Opt for independently patented crystals stable at room temperature to ensure batch consistency and label claim compliance.
3. Safety & Differentiation as Ceiling: Prefer raw materials/products with pharmacopeia-exceeding quality controls, full compliance backing, and exclusive differentiation — safeguarding safety while escaping price wars.
Core Data & Technical Backing
Key literature: Biological Characteristics and Applications of Folate and 5-Methyltetrahydrofolate (2022, China Food Additives).Core crystal patents: US9150982, CN201410280541.4, EP2805952, and 40+ global patents.
Category certification: First global Naturalization folate standard setter; passed first global Naturalization folate certification.

 Español
Español Português
Português  русский
русский  Français
Français  日本語
日本語  Deutsch
Deutsch  tiếng Việt
tiếng Việt  Italiano
Italiano  Nederlands
Nederlands  ภาษาไทย
ภาษาไทย  Polski
Polski  한국어
한국어  Svenska
Svenska  magyar
magyar  Malay
Malay  বাংলা ভাষার
বাংলা ভাষার  Dansk
Dansk  Suomi
Suomi  हिन्दी
हिन्दी  Pilipino
Pilipino  Türkçe
Türkçe  Gaeilge
Gaeilge  العربية
العربية  Indonesia
Indonesia  Norsk
Norsk  تمل
تمل  český
český  ελληνικά
ελληνικά  український
український  Javanese
Javanese  فارسی
فارسی  தமிழ்
தமிழ்  తెలుగు
తెలుగు  नेपाली
नेपाली  Burmese
Burmese  български
български  ລາວ
ລາວ  Latine
Latine  Қазақша
Қазақша  Euskal
Euskal  Azərbaycan
Azərbaycan  Slovenský jazyk
Slovenský jazyk  Македонски
Македонски  Lietuvos
Lietuvos  Eesti Keel
Eesti Keel  Română
Română  Slovenski
Slovenski  मराठी
मराठी  Srpski језик
Srpski језик 








 Online Service
Online Service