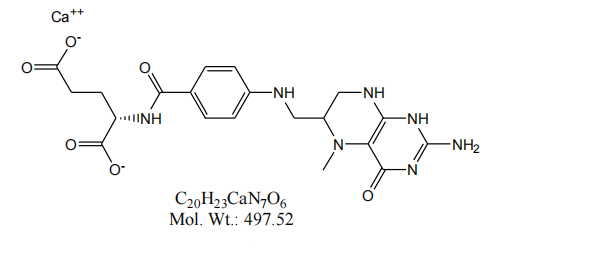
Calcium L-5-methyltetrahydrofolate is an important nutritional supplement that is widely used in the nutraceutical and pharmaceutical industries. Product quality testing is critical for the manufacturer as it is directly related to the safety and effectiveness of the product. This article will describe the characteristics of L-5-methyltetrahydrofolate calcium and focus on how manufacturers should conduct quality inspection.
Quality inspection of L-5-methyltetrahydrofolate calcium is very important to the manufacturer. First and foremost, manufacturers need to ensure that the raw materials they purchase are a compliant, high quality product. The quality of ingredients directly affects the quality and effectiveness of the final product. Therefore, manufacturers should purchase raw materials from reliable and reputable suppliers and require suppliers to provide appropriate quality documentation.
Secondly, manufacturers should establish a strict production process and quality control system. Each step of the production process should be monitored and inspected to ensure the stability and consistency of the products. Manufacturers can use advanced technology and instruments to monitor key parameters in the production process, such as temperature, humidity, pH, etc., to ensure the quality of the products.
Third, manufacturers should also conduct quality inspection of finished products. This includes checking the appearance, physical and chemical properties and purity of the product and other indicators. The manufacturer can commission an independent third-party laboratory to conduct the testing to ensure the objectivity and accuracy of the test results. At the same time, manufacturers can also develop their own quality standards and testing methods to meet the needs of different markets and customers.
In addition, manufacturers should establish a sound quality management system. This includes training employees, developing standard operating procedures, and establishing a defective product management and traceability system. Manufacturers should continuously improve the quality management system to continuously improve the quality and competitiveness of their products.
In conclusion, quality inspection of calcium L-5-methyltetrahydrofolate is crucial for manufacturers. Manufacturers should strictly follow the relevant standards from raw material procurement to finished product quality inspection. Only by ensuring product quality can manufacturers win the trust of customers and competitive advantage in the market. Therefore, manufacturers should pay high attention to quality inspection and continuously improve and refine the quality management system in order to provide high quality L-5-methyltetrahydrofolate calcium ingredient.

The Magnafolate® calcium L-5-methyltetrahydrofolate is safer, purer, more stable and suitable for a wide range of people including those with MTHFR gene mutations. Calcium L-5-methyltetrahydrofolate does not need to be metabolized in the body and can be absorbed directly.
Email: info@magnafolate.com

 Español
Español Português
Português  русский
русский  Français
Français  日本語
日本語  Deutsch
Deutsch  tiếng Việt
tiếng Việt  Italiano
Italiano  Nederlands
Nederlands  ภาษาไทย
ภาษาไทย  Polski
Polski  한국어
한국어  Svenska
Svenska  magyar
magyar  Malay
Malay  বাংলা ভাষার
বাংলা ভাষার  Dansk
Dansk  Suomi
Suomi  हिन्दी
हिन्दी  Pilipino
Pilipino  Türkçe
Türkçe  Gaeilge
Gaeilge  العربية
العربية  Indonesia
Indonesia  Norsk
Norsk  تمل
تمل  český
český  ελληνικά
ελληνικά  український
український  Javanese
Javanese  فارسی
فارسی  தமிழ்
தமிழ்  తెలుగు
తెలుగు  नेपाली
नेपाली  Burmese
Burmese  български
български  ລາວ
ລາວ  Latine
Latine  Қазақша
Қазақша  Euskal
Euskal  Azərbaycan
Azərbaycan  Slovenský jazyk
Slovenský jazyk  Македонски
Македонски  Lietuvos
Lietuvos  Eesti Keel
Eesti Keel  Română
Română  Slovenski
Slovenski  मराठी
मराठी  Srpski језик
Srpski језик 








 Online Service
Online Service